One Step Test for SARS-CoV-2 Antigen
(Colloidal Gold) (Nasal Swab)
- For self-test
Intended Use
One Step Test for SARS-CoV-2 Antigen (Colloidal Gold) is intended for the qualitative detection of SARS-CoV-2 antigens in human nasal swab samples.
This test is suitable for medical laypersons as a self-test at home or at work.
About SARS-CoV-2
The novel coronavirus belong to the β genus. SARS-CoV-2 is an acute respiratory infectious disease. People are generally susceptible. Currently, the patients infected by the novel coronavirus are the main source of infection; asymptomatic infected people can also be an infectious source. Based on the current epidemiological investigation, the incubation period is 1 to 14 days, mostly 3 to 7 days. The main manifestations include fever, fatigue and dry cough. Nasal congestion, runny nose, sore throat, myalgia and diarrhea are found in a few cases.
Use this test:
- If you want to test yourself.
- If you have symptoms similar to COVID-19, such as headache, fever, cough, sore throat, loss of the sense of smell or taste, shortness of breath, muscle pain.
- If you are concerned about whether you are infected with SARS-CoV-2.
- Use of the test by persons under 18 years of age only under the supervision of an adult.
Contents
A kit contains:
Package specifications: 1 T/kit, 5 T/kit, 7 T/kit, 25 T/kit
1) SARS-CoV-2 antigen test card
2) Extraction tube with sample extraction solution and tip
3) Sterile swab
4) Biohazard sample bag
5) User manual: 1 piece/kit
Additional required material: clock/ timer/ stopwatch
Note: Do not mix or interchange different batches of kits.
Specifications
|
Test Item |
Sample Type |
Storage Condition |
|
SARS-CoV-2 Antigen |
Human nasal swab |
4-30℃ |
|
Methodology |
Test Time |
Shelf Life |
|
Colloidal Gold |
10-15 min |
24 months |
Operation
Nasal Swab Sampling
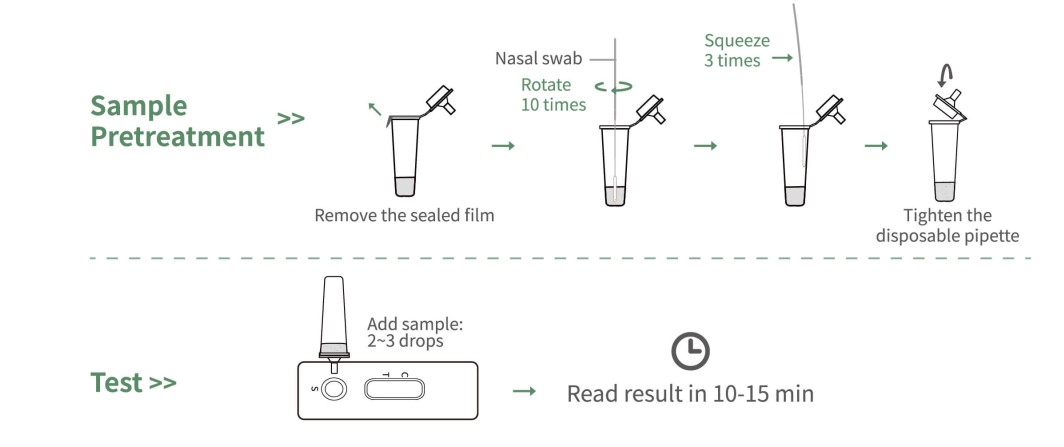
Test Results
1. Valid Test
Positive (+):
Two bands appear, one at the control area (C) and the other at the test line (T). The result indicates the presence of SARS-CoV-2 antigen.
Negative (-):
A single band appears at the control area (C) and no other band at test line. The result indicates that the sample does not contain SARS-CoV-2 antigen or the concentration is below the limit of detection of the kit.
2. Invalid Test
If no band appears in the control area (C), the test result is invalid. The test should be repeated with a new test card and if the same situation reappears, please stop using this batch of products and contact your supplier.